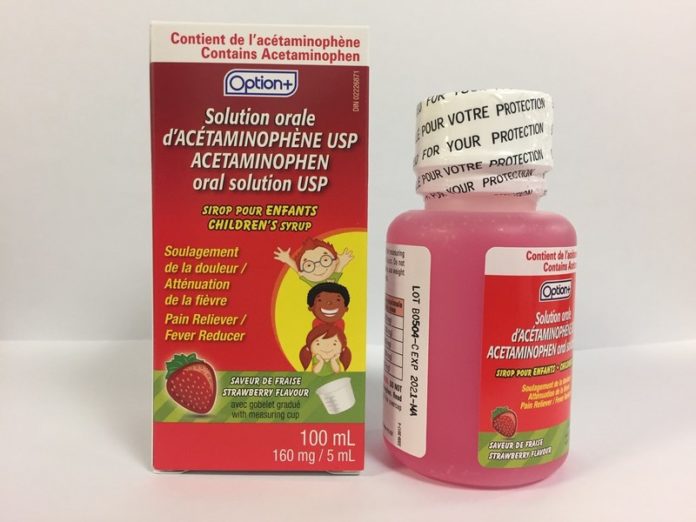
Health Canada has issued a recall for several children’s strawberry-flavoured acetaminophen syrups for a defective child-resistant lids.
The product is used for pain and fever relief, and while nothing is wrong with the syrup itself, the defective child-resistant lid could be potentially dangerous for children.
Because of the faulty packaging, Laboratoire Riva Inc. and Laboratoires Trianon Inc. are voluntarily recalling several of their over-the-counter children’s medicinal syrups labeled as Biomedic, Option+, or Laboratoires Trianon Inc.
A defective cap could allow a child to accidentally ingest the product, which could result in acetaminophen overdose or serious health consequences, including liver damage in extreme cases.
If you are concerned about overdose, signs include nausea, vomiting, lethargy, and sweating. If untreated, liver damage may occur and possibly result in liver failure or death. Abdominal pain may also be the first sign of liver damage and could appear 24 to 48 hours after ingestion.
If you think your child may have taken too much acetaminophen, call your local poison control centre immediately.
What to do if you have one of these products
- You can return the affected product to the place of purchase for a refund.
- Store medications out of reach and sight of children.
- Report any health product-related adverse reactions or complaints to Health Canada.
- If you have questions about the recall you can contact Urszula Wiatrowska, a pharmacovigilance specialist at Laboratoire Riva and Laboratoires Trianon Inc.,
Email uwiatrowska@labriva.com, or call toll-free at 1-800-363-7988, extension 236.